Activity Implementation Report
Wei Group PhD Student
PhD Year 2 Yue Xu
Affiliation: Graduate School of Medicine, Tohoku University / Dept. Modomics Biology & Medicine, Tohoku University
Host Institution: Massachusetts General Hospital, USA, Liron Bar-Peled Laboratory
Research Topic: Elucidation of cellular function regulatory mechanisms through high-throughput CRISPR screening


From October to December 2025, for approximately three months, I conducted a short-term overseas research training program in the laboratory of Liron Bar-Peled at Massachusetts General Hospital / Harvard Medical School in the United States. The Bar-Peled laboratory is a world-leading research group that focuses on the intersection of cellular metabolism and signal transduction, and aims to elucidate how cancer cells respond and adapt to changes in metabolic states using cutting-edge molecular biology approaches such as CRISPR screening and chemical proteomics.
During my stay, I performed pooled CRISPR screening using the U937 cell line, and systematically learned the entire series of experimental and analytical processes, from sgRNA library preparation and management of cell coverage to data acquisition and initial analysis using next-generation sequencing (NGS). In addition, using one of the chemical proteomics techniques established in this laboratory—cysteine chemical proteomics—I conducted analyses of protein structural changes associated with compound binding through DBIA enrichment experiments.
Furthermore, through discussions with laboratory members, I learned practical approaches to interpreting screening data, prioritizing candidate genes, and designing follow-up experiments. Conducting the entire process from experimental system setup to execution and data interpretation within the limited period of approximately three months was a significant challenge; however, I feel that this experience allowed me to acquire both the mindset and execution ability necessary to drive research forward
The knowledge and experimental foundations obtained through this overseas training are being directly applied to my research after returning to Japan. Currently, I am advancing detailed molecular mechanism analyses of targets identified during the training period, while also formulating plans for future research development. Through this short-term overseas research program, my ability to independently conduct research in an international research environment has been greatly enhanced, and I was able to establish an important foundation for my future research activities.
I would like to express my sincere gratitude to the International Leading Research Program for its generous support of this research, and I would also like to extend my heartfelt thanks to all members of the Bar-Peled laboratory for their warm hospitality.
Akaike Group Undergraduate Student
Zizai Shen
Undergraduate Year 3
Affiliation: School of Medicine, Tohoku University / Graduate School of Medicine, Tohoku University, Department of Redox Molecular Medicine (Prof. Takaaki Akaike’s Laboratory)
Host Institution: Max Planck Institute for Polymer Research, Laboratory of Dr. Uladzimir Barayeu (Mainz, Germany)
Research Topic: Co-translational protein modification mediated by cysteine persulfidation activity of CARS
(As of January 2026)



I conducted a short-term research stays at the Max Planck Institute for Polymer Research to investigate co-translational protein modification mechanisms mediated by cysteine persulfidation. Previous studies suggest that cysteinyl-tRNA synthetase (CARS), in addition to its canonical aminoacylation activity, catalyzes cysteine persulfidation, suggesting its involvement in co-translational protein modification. My research focused on whether CARS2 directly charges cysteine persulfide onto tRNA or whether cysteine is first aminoacylated and subsequently persulfidated.
Specifically, I examined human and mouse CARS2 proteins carrying mutations in the KMSK and KIIK motifs, which have been implicated in persulfidation activity. Using a bacterial expression system, I optimized the conditions for protein expression and purifications, enabling the isolation of mouse CARS2 with sufficient purity and yield for functional studies. In this study aboard program, I acquired to design experiments, validate hypothesis, optimize of experimental conditions, and interpretate experimental data. I also gained confidence that the molecular biology and protein biochemistry techniques acquired in Japan are fully applicable in an international research environment. Moreover, engaging in scientific discussions in English significantly broadened my perspective as a researcher.
The findings obtained during this study will be further developed toward elucidating the molecular mechanisms underlying co-translational protein modification mediated by cysteine persulfidation. I intend to fully leverage the experience and outcomes gained through this International Leading Program in my future research activities.
Miki Group PhD Student
Graduate School of Engineering, Kyoto University (Miki lab)
Koyuki Kawamura, Second grade in doctor course
Helmholtz Zentrum München, Dr. Marcus Conrad lab (Germany)
July ~ October, 2024
Elucidation of the cell death mechanism induced by intracellular Mg²⁺ decrease


From July to October 2024, I had a chance to conduct research abroad for three months at the Marcus Conrad lab, Helmholtz Zentrum München (Germany). The Conrad lab is a globally leading research group in the field of ferroptosis, a form of programmed cell death. I had found that a decrease in intracellular Mg²⁺ levels induces a unique mode of cell death, which provided me this great opportunity to extend my research.
During my stay there, I established a cell line, of which intracellular Mg²⁺ levels can be inducibly lowered, using HT-1080 cells that are widely used in cell death research. Furthermore, I performed time-lapse imaging of cell death using a holographic microscope, a label-free technique enabling high-magnification and high-resolution observation. As a result, I succeeded in recording instantaneous and dynamic morphological changes that had previously eluded conventional microscopy. This result revealed that the dying cells induced by intracellular Mg²⁺ decrease exhibit a unique behavior that cannot be categorized under any known form of cell death. Viewing the movies with Dr Conrad and colleagues in the microscopy room, where we had very lively discussions, remains one of my cherished memories from my time abroad.
Including these findings, we received guidance from the Conrad lab regarding the unique cell death induced by Mg²⁺ decrease and its mechanisms, leading to the submission of our paper. Furthermore, we have conducted screening analyses using cell lines established during my overseas research period and are advancing studies aimed at elucidating further mechanisms.
During this period, I faced considerable pressure to achieve tangible results within the limited three-month timeframe. However, by maintaining a nimble approach to experimentation through daily trial and error, I was able to return home with intriguing findings. This experience has instilled significant confidence in my future research endeavors.
I would like to express my sincere gratitude once again to the International Leading Research program for supporting this opportunity, and to all members of the Conrad lab for their warm hospitality.
Motohashi Group PhD Student
Haruna Takeda, 4th-year PhD student (at the time)
Affiliation (at the time): Department of Medical Biochemistry, Tohoku University Graduate School of Medicine (Principal Investigator: Dr. Hozumi Motohashi)
Host institution: Department of Cancer Biology, Dana-Farber Cancer Institute (Principal Investigator: Dr. Edward Chouchani)
Research title: Physiological and Druggble Redox Signaling Governs T cell Activation


From July 2024 to March 2025, I visited Dr. Edward Chouchani’s lab at the Dana-Farber Cancer Institute as a visiting student. The Chouchani lab has developed a novel mass spectrometric method called “CPT-based redox proteomics,” which enables stoichiometric quantification of reversible cysteine oxidations on a proteome-wide scale.
T cells, key players in adaptive immunity, produce redox-active metabolites such as reactive oxygen species and hydrogen peroxide upon antigen stimulation. These metabolites are known to play essential roles in regulating T cell function, but the underlying molecular mechanisms remain unclear. During my visit, I performed CPT-based redox proteomics on human and mouse CD4⁺ and CD8⁺ T cells to identify cysteine residues that undergo oxidative modification following antigen stimulation, aiming to discover novel redox sensors that regulate adaptive immunity. The results revealed dynamic changes in the oxidation state of numerous cysteine residues during T cell activation.
I am now focusing on several of these cysteine residues to elucidate the functional significance of oxidative modifications in T cell function. I am also working on developing covalent drugs that selectively modify these cysteine residues, which could potentially be applied as novel therapeutic approaches against cancers and immune-related diseases. One future goal is to extend CPT-based redox proteomics to quantify supersulfide modifications.
To continue this research, I applied to several fellowships during my stay and was fortunate to be awarded a Long-Term Fellowship from the Human Frontier Science Program (HFSP). Starting this April, I have been working as a Postdoctoral Fellow at the Chouchani lab. I will devote myself to expanding this research, which was initiated with support from this International Leading Research Grant.
Lastly, I would like to express my sincere gratitude for this invaluable opportunity and to all those who have contributed to and supported this work.
[Achievements]
- H. Takeda, et al., “OxiT: A Comprehensive Stoichiometric Landscape of Cysteine Oxidation During T Cell Activation”. Redox Week in Sendai, Sendai, Japan. Apr 2025.
- H. Takeda, et al., “Mediator Complex Subunit 16 (MD16): Its Physiological Role and Redox-dependent Regulations” Gordon Research Seminor/Conference (Thiol-Based Redox Regulation and Signaling), Barcelona, Spain. July 2024.
- Ikushi Award, JSPS
- Long-Term Fellowship, Human Frontier Science Program
Nakabayashi Group PhD Student
Keisuke Koga (Graduate School of Pharmaceutical Sciences, Tohoku University)


From April to June 2024 (two months), I conducted research as a visiting student in the laboratory of Associate Professor Hirotsugu Hiramatsu at National Yang Ming Chiao Tung University, Taiwan. During this period, I developed novel Raman spectroscopy-based detection methods for supersulfide molecules. In particular, I focused on the highly sensitive detection of supersulfide molecules by combining the Vertical Flow (VF) Raman method with high-performance liquid chromatography (HPLC), enabling high-sensitivity HPLC–Raman measurements.
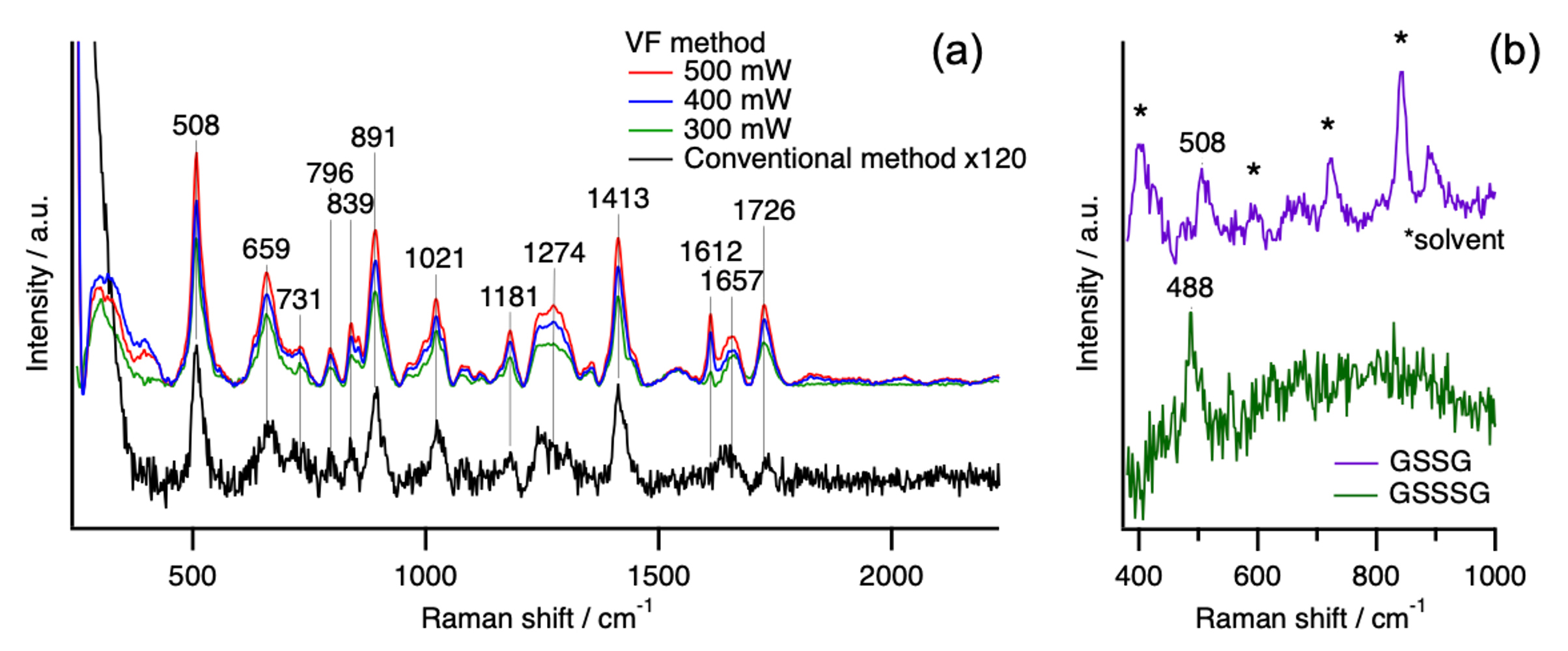
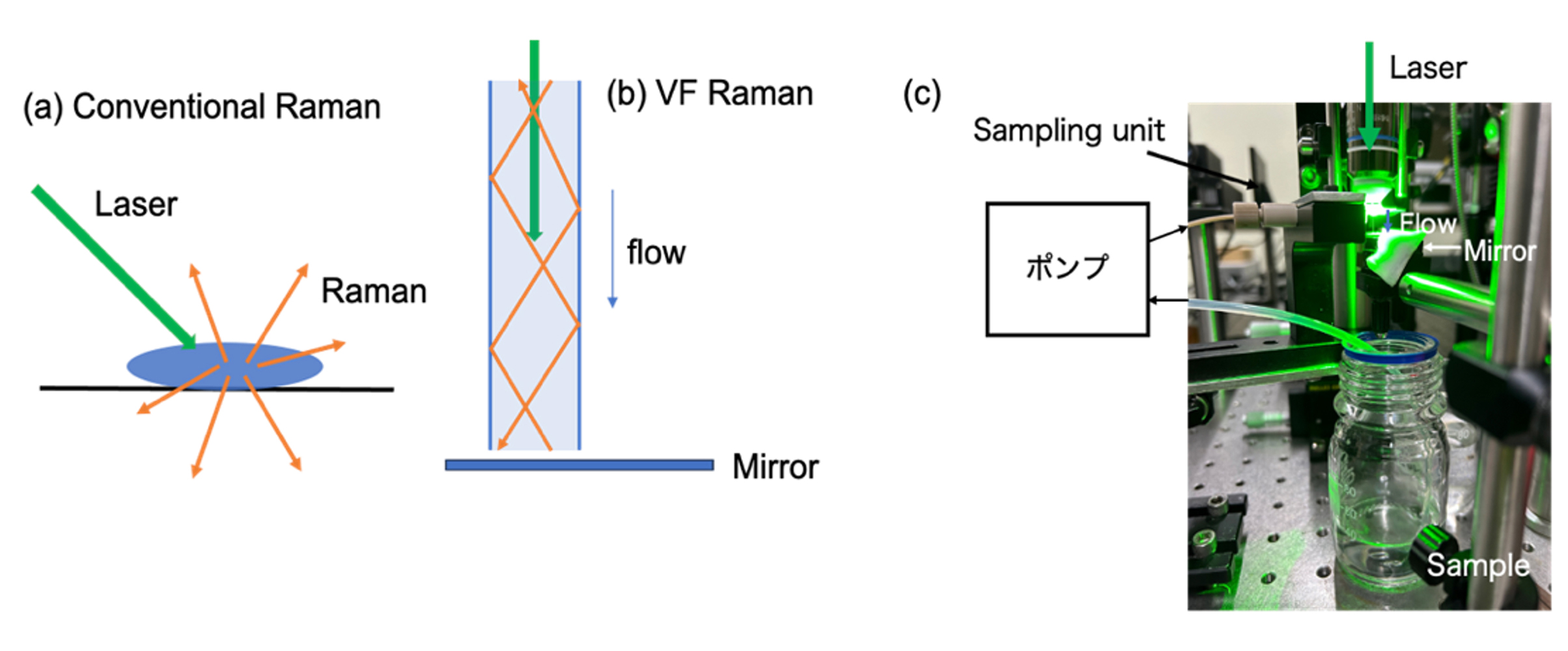
The VF method enhances Raman intensity by repeatedly reflecting the excitation light within a water column through total internal reflection (Fig. 1b). Using an aqueous solution of oxidized glutathione (GSSG), we evaluated the signal enhancement and successfully achieved approximately a 120-fold increase in the intensity of the S–S Raman band compared with the conventional method (Fig. 2a).
Next, we separated and detected a mixed solution of GSSG and oxidized glutathione trisulfide (GSSSG) using LC–Raman spectroscopy, which combines HPLC with VF Raman detection. The LC–Raman system enabled clear separation of GSSG and GSSSG, and Raman spectra obtained for each component allowed the distinction between Raman bands due to S–S and S–S–S (Fig. 2b). In addition, Raman spectra of sulfur molecules were also measured using a novel technique known as hyper-Raman spectroscopy.
In future experiments, we plan to measure supersulfide molecules other than GSSSG and compile the results into a research paper. We also aim to investigate the detection of supersulfide molecules as reactive species, such as reactions between Na₂S₄ and organic molecules, using LC–Raman spectroscopy. The VF-based LC–Raman method offers significantly higher sensitivity than conventional LC–Raman methods, and we aim to establish it as a versatile, general method for supersulfide detection.
[Presentation] 1. K. Koga, et al., Development of a new detection method for supersulfide molecules using Raman scattering. Redox Week in Sendai 2025.
Akaike Group Ph D Student
Name: Masana Yazaki
Academic Status: Third-Year Ph.D. Student
Affiliation: Arisawa Laboratory, Faculty of Agriculture, Kyushu University
Host Institution: Brown University, Rhode Island, USA
Host Researcher: Prof. Ming Xian
Period of Stay: September 1 – October 5, 2024
Research Theme: Chemical Synthesis of Redox Signaling Molecule Donors



During my research stay in Prof. Ming Xian’s laboratory at the Brown University, I was engaged in the design and synthesis of nitrosothiol (SNO) compounds capable of the controlled release of both nitric oxide (NO) and hydrogen sulfide (H₂S). These dual donors release NO upon photoirradiation to generate disulfides, which subsequently liberate H₂S in response to specific chemical stimuli. By fine-tuning the molecular framework, we aimed to precisely regulate the timing and interplay of NO and H₂S release, thereby providing promising chemical tools for elucidating redox signaling mechanisms. Although development of a synthetic route that prevented premature release of the active species was initially challenging, intensive discussions with Prof. Xian and laboratory members enabled me to successfully achieve efficient preparation of the target molecules within the one-month period. The results of this work are currently being submitted to an international journal.
In our laboratory, I am engaged in the development of transition-metal-catalyzed transformations of S–S bonds in peptide polysulfides. The experience and technical insights gained during my stay abroad are highly helpful for my ongoing project to develop innovative strategies for the controlled release of reactive sulfur and nitrogen species without photoirradiation. This valuable international opportunity significantly promoted my doctoral research in peptide polysulfides chemistry.
I would like to express my deepest gratitude to Prof. Akaike and all those involved in these Program for their generous support of my research stay in the United States.
Wei Group Master’s Student
Visiting Scholar: Yusuke Okubo, 1st-year Master's Student, Graduate School of Life Sciences, Tohoku University
Period: September 2024 - January 2025
Visited Institution: Yale School of Medicine, Elizabeth Laboratory (Photo below: Professor Elizabeth)


Purpose: At the Elizabeth Laboratory in the Department of Endocrinology, Yale School of Medicine, purify submitochondrial particles (SMP) from rat liver using centrifugation. Subsequently, at the laboratory of Assistant Professor Jack Zhang at the same university, observe the purified samples using cryo-electron microscopy single particle analysis (SPA) and cryo-electron tomography (Cryo-ET), and perform 3D reconstruction. The objective is to elucidate the molecular identity of the mitochondrial permeability transition pore (mPTP), a complex comprising the adenine nucleotide transporter (ANT), VDAC, and cyclophilin D subunits, from a structural biology perspective.
Results: We successfully purified the target protein and acquired data using cryo-electron microscopy. SPA yielded approximately 24,000 microscope images, while Cryo-ET produced data from 115 tilt series. These data were stored on an HDD, which was brought back to Japan.
Achievements: Using SPA, we successfully reconstructed the 3D structure of ATP synthase at 3.62 Å resolution.
Prospects: To clarify the molecular identity of the mPTP, it is necessary to proceed with analysis using the currently available data. Regarding cryo-ET, further study and verification of analysis methods are required, which we intend to pursue in the future.